P-ISSN: 2394-0530, E-ISSN: 2320-3862
2016, Vol. 4, Issue 3, Part A
Phytochemicals and acute oral toxicity studies of the aqueous extract of Vernonia amygdalina from state of Malaysia
Yusmazura Zakaria, Nurhazirah Zainul Azlan, Nik Fakhuruddin Nik Hassan, Hussin Muhammad
Vernonia amygdalina (VA) is a member of Asteracea family that use as traditional folk medicine in treating various infection and diseases. This study aim to determine the phytochemical characterization of Malaysian local VA and determine the acute oral toxicity. Phytochemical investigation was done by standard procedures. The crude extract of VA was observed to contained flavonoids, terpenoids, saponin and tannin. FTIR spectroscopy was done against VA aqueous extract revealed a presence of high content of flavonoids and terpenoids, phenols, methoxy compounds, ester carbonyl and amide. Sighting study for acute toxicity was conducted as per Organization of Economic Cooperation and Development (OECD) 425 guidelines which divided into sighting study and main study. One female Sprague Dawley rat was given single oral dose of VA aqueous extract at progressively dose of 175, 550, 2000 and 5000 mg/kg of body weight (BW), dissolved in 1ml distilled water and observed daily for 14 days. VA aqueous extract of 5000 mg/kg was used in main study as it exhibited no toxicity signs and mortality. All the treated rats survived and no toxicity signs were observed. No differences in body weights and organs weights. Liver function tests showed the levels of aspartate aminotransferase (AST) and globulin slightly increased but the levels of alanine aminotransferase (ALT), gamma-glutamyl transferase (GGT), albumin, total protein and total bilirubin were in normal range. Despite that, no elevation in the level of Alkaline phosphatase (ALP). Therefore, it can be concluded that the toxicity of VA is greater than 5000mg/kg BW as proved by AOT425 Statistical Programme.
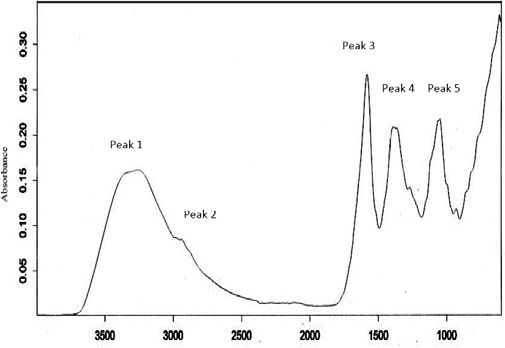
Fig.: ATR-FTIR spectrum of V. amygdalina
Pages : 01-05 | 2024 Views | 371 Downloads
How to cite this article:
Yusmazura Zakaria, Nurhazirah Zainul Azlan, Nik Fakhuruddin Nik Hassan, Hussin Muhammad. Phytochemicals and acute oral toxicity studies of the aqueous extract of Vernonia amygdalina from state of Malaysia. J Med Plants Stud 2016;4(3):01-05.
Related Journal Subscription
Important Publications Links






